Determination of Fatty Acid Methyl Esters in olive oil using GC-SQMS
Introduction
The determination of Fatty Acid Methyl Esters (FAME) is a commonly performed analysis, to determine the quality of extra virgin olive oil (EVOO). Natural EVOO is made by pressing or centrifuging olives, without exposing the olives to any chemical processing. A way of producing cheaper olive oil is to mix them with cheaper oils, such as sunflower oil and refined olive oils. This mixing with refined olive oils results in a divergent fatty acid content in the end product.
The determination of fatty acids for quality control in EVOO’s is performed by Gas Chromatography (GC). Therefore, the fatty acids need to be trans esterified into FAME because of the thermally stable volatile behaviour of these compounds. This results in a more reliable analysis. In the transesterification process free and bonded fatty acids react with an alcohol in the presence of a catalyst, forming a mixture of FAME compounds and an alcohol.1 The composition of FAME compounds in EVOO can say much about the quality and origin of the olive oil, which makes this analysis very valuable for quality control.
| This application will study the quantification and qualification of FAME within EVOO’s, by GC with Mass Spectrometry(MS).
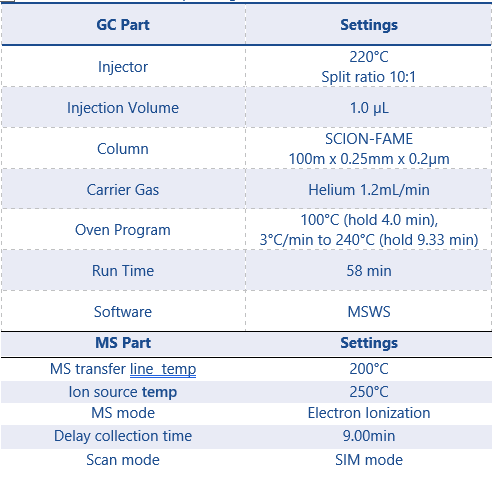
This application can be performed on either the SCION Instruments 8300 GC & 8500 GC platform with 8700 Single Quad Mass Spectrometer (SQMS) and the SCION 8400PRO Autosampler, shown in Figure 1. A SCION-FAME column is used for obtaining the best separation of the cis and trans-isomers of most FAMEs. |

Figure 1 SCION Instruments 8300 & 8500-GC and 8700 SQMS and 8400PRO Autosampler.
Table 1 details the GC and SQMS method parameters used throughout this analysis.
Table 1: Instrumentation operating conditions GC and SQMS
Experimental
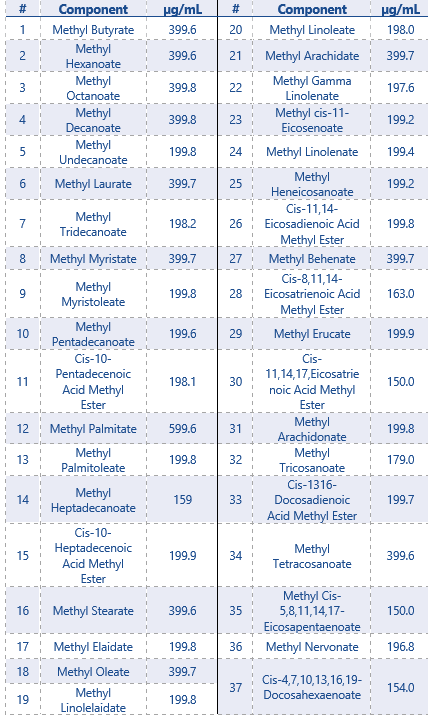
For this application a FAME standard was purchased for the qualification and quantification of unknown samples.
The FAME standard contained 37 FAME compounds with a concentration range from 150 – 600 µg/mL.
The components present in the FAME standard are shown in Table 2.
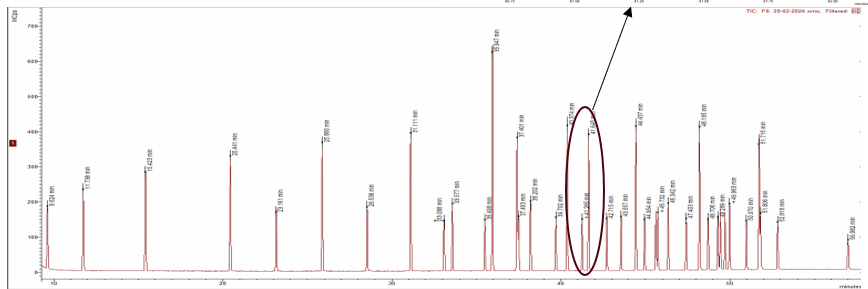
The SCION-FAME column provides a perfect resolution between the cis and trans FAME isomers that are present in the FAME-mix, seen in the chromatogram in Figure 3. Therefore all 37 FAME components were successfully identified with NIST library search tool built into the MS Work Station (MSWS) software. The method was easily converted from Full scan into a SIM method, which increases the sensitivity of the method.
For example, compound #1 Methyl Butyrate is identified with the NIST library. When this component is added to the component list assigned to the method, the Quantifier (74 m/z) and Qualifier ions (43 and 71 m/z) are also inserted into the method. The retention time match and match with the known mass spectrum makes it very easy to identify unknown samples.
Table 2: Components and concentrations of the FAME-standard
Sample Preparation
A calibration set from the FAME standards (diluted with n-Heptane) prepared from 0.20 µg/mL up to 50 µg/mL. Deuterated Methyl Heptadecanoate (50 µg/mL) is used as an internal standard (IS) and added to all standards and samples. An IS working solution 1000 µg/ml is prepared by dissolving 10 mg Deuterated Methyl Heptadecanoate into n-heptane, in a 10 mL volumetric flask.
To prepare the EVOO sample, 0.1 g of the olive oil is transferred into a test tube. To this tube 250 µL of the IS work solution is added plus 4750 µL n-Heptane, followed by shaking. Then 200 µL of 2M KOH dissolved in methanol is added to perform the esterification process, by shaking vigorously for 30 seconds. The solution is left to stratify until the upper layer of the solution becomes clear. The upper layer is transferred into a GC sample vial and is ready for injection.2
Blank injections of n-Heptane are performed in between samples, to ensure that the system is not contaminated after sample injections.
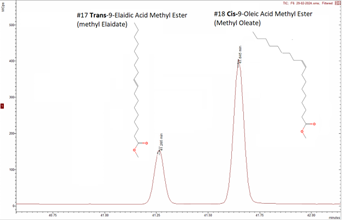
Figure 2 TIC chromatogram of the FAME-standard
Results
| Due to the number of components present in this standard and method, not all compounds will be mentioned in the Results section.
If necessary, the full validation report is accessible by request. The results for the following FAME compounds found in the EVOO sample are shown: Methyl Palmitate (#12), Methyl Palmitoleate (#13), Methyl Stearate (#16), Methyl Oleate (#18), Methyl Linoleate (#20), Methyl Arachidate (#21), and Methyl Linolenate (#24). The calibration curves for the FAME standards were prepared from 0.2 µg/mL up to 50 µg/mL. The precision of the method was obtained by seven consecutive injections of FAME standard #4 (range 10-25 µg/mL). |
The results of the precision of the selected FAME compounds can be found in Table 3, along with the linearity results (R2) obtained by the calibration curves.
Table 3: Summary of Results – Linearity and repeatability
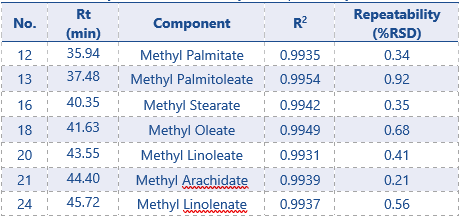
For all FAME compounds an R2 of 0.99 or higher was achieved. This is an excellent result, with many regulations requiring an R2 value of ≥0.98. Repeatability results show that for most FAME compounds the relative standard deviations (RSD%) are below 1%. 2 compounds have an RSD ≤1.5%. These results show a good system precision for this method, since most acceptance criteria for method validation are requiring an RSD ≤2%.
The limit of detection (LOD) and limit of quantitation (LOQ) were calculated according to equations 1 and 2:

| Where the noise (in kCps) is calculated from a blank injection, the concentration of the components is from calibration standard #1 (800x dilution of FAME standard, in µg/mL) with corresponding peak height (in kCps).
The calculated LOD’s were found to be ≤0.04 µg/mL, these are shown in Table 4. These low LOD and LOQ’s are easily achievable when measured in SIM-mode. |
Identified FAME compounds found in the sample are shown in Table 4. According to regulations an EVOO FAME content (summation of total FAMEs) should be ≤75 mg/kg.3 If compensating for the dilution in n-Heptane the Ʃ FAME content this is ≤51.3 µg/mL. In the olive oil sample 7 FAME compounds are identified in MSWS, the concentration of each component is calculated from the calibration curves equations. The total Ʃ FAME-content from all identified components is 39.96 µg/mL, and therefore within the specification mentioned in EU regulations.
Table 4: Summary of Results – LOD, LOQ and Amount found in extra virgin olive oil sample.
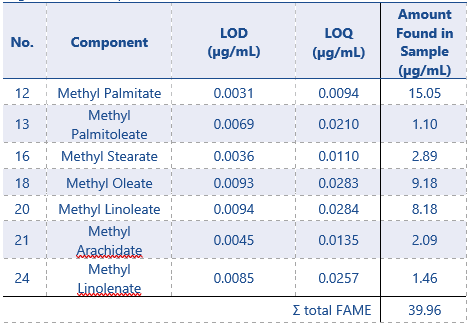
Figure 3 shows some example chromatograms from MSWS of the target peaks Methyl Oleate and Methyl Linoleate and their corresponding mass spectra, from the EVOO sample. Also shown in Figure 3 is the NIST library reference mass spectra confirming the identity of each analyte.
Also shown in Figure 3 is the NIST library reference mass spectra confirming the identity of each analyte.
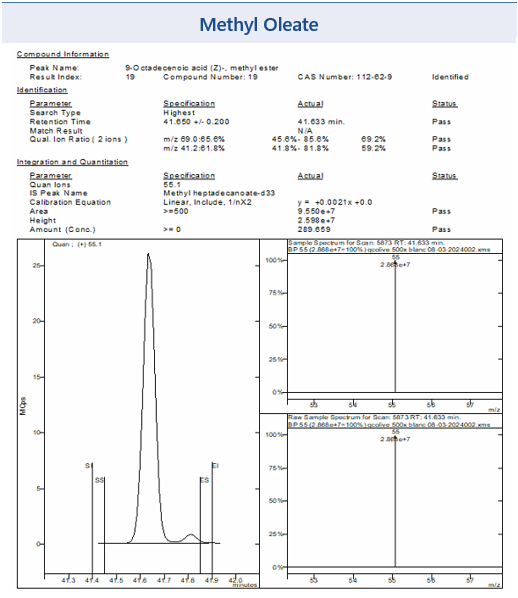
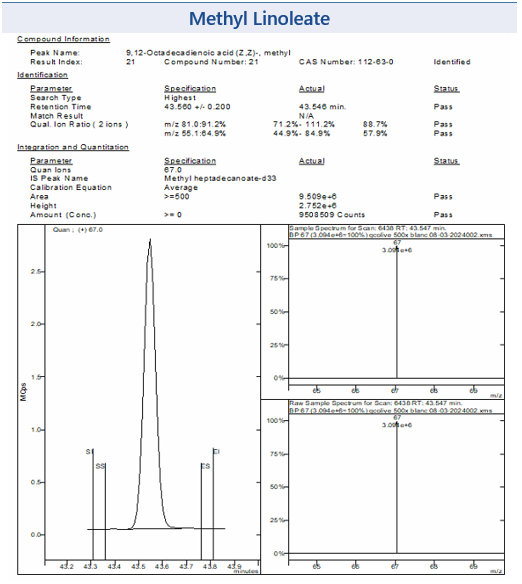
Figure 3: Example chromatograms for selected FAMEs
Extra virgin olive oils are known for their health benefits, derived from a high content of Methyl oleate and a lower content Methyl Linoleate.
Table 5: Results FAME composition in EVOO sample compared to specifications mentioned in regulations4
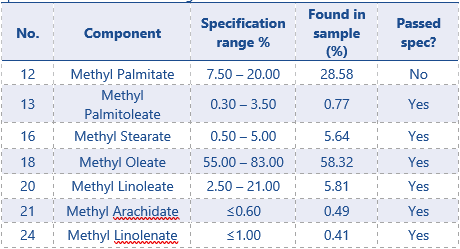
The composition range (%) of FAMEs mentioned in regulations4 for EVOO are shown in table 5.
The composition of identified FAMES from the EVOO sample (% peak Areas) are compared with the set up specifications. Only 1 of the 7 FAME compounds is above spec (methyl palmitate). When a EVOO is suspected to be adulterated, this mostly results in a divergent Methyl Oleate or Methyl Linoleate contents, not for methyl palmitate.
Another confirmation that be sure an EVOO is not adulterated is to check the presence of trans-isomers. Trans-isomers (for example Methyl Elaidate) are double bonded fatty acids which are not present in natural EVOO, but can be formed after heating processes. In this case the analyzed EVOO is not containing any trans-FAME compounds which could indicate that the sample is not exposed to any heating processes.
With the total FAME content mentioned in table 4, the composition of FAME components in table 5, and no presence of trans-isomers in the sample it can be assumed that the analyses sample is an genuine EVOO, that is most likely not exposed to dilution with cheaper oils, or any chemical processes.
Conclusion
The SCION 8500 GC platform equipped with a split/spitless injector, SCION FAME column and 8700 SQMS and 8400PRO sampler is a perfect solution for analyzing FAME in olive oil for qualitative and quantitative analysis. A good system precision, good linearity results and low LOQ’s and LOD’s are achieved for this application with the SCION Instruments GC-MS set up and MS-Work Station software.
The SCION-FAME column shows good resolution between cis and trans FAME isomers, which is an easy way to verify if there are no trans-FAME isomers present in an EVOO that could indicate heating in the production process.
The analyzed EVOO sample is, according to multiple results obtained by this application, most likely a natural EVOO that has not been exposed to adultery in the production process.
This method is also applicable on the SCION Instruments 8300 or 8500 GC-platform with Flame Ionisation Detector (GC-FID).
References
1. INTERNATIONAL OLIVE COUNCIL; DETERMINATION OF FATTY ACID METHYL ESTERS BY GAS CHROMATOGRAPHY, https://www.internationaloliveoil.org/wp-content/uploads/2019/11/COI-T.20-Doc.-No-33-Rev.-1-2017.pdf (accessed 23 Feb 24)
2. COMMISSION IMPLEMENTING REGULATION (EU) Characteristics of olive oil and olive-residue oil and on the relevant methods of analysis 2015, https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32015R1833&rid=8 (accessed 14 Feb 24)
3. COMMISSION REGULATION (EU) Characteristics of olive oil and olive-residue oil and on the relevant methods of analysis 2011, https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32011R0061 (accessed 30 Apr 25)
4. INTERNATIONAL OLIVE COUNCIL: TRADE STANDARD APPLYING TO OLIVE OILS AND OLIVE POMACE OILS JUNE 2019 , https://www.internationaloliveoil.org/wp-content/uploads/2019/11/COI-T.15-NC.-No-3-Rev.-13-2019-Eng.pdf (accessed 29 Mar 24)
Download The Application Note
Download the application note: AN168v2 Determination of FAMEs in olive oil using GC-SQMS