Residual Solvent Analysis of Chemical Products by GC-FID with Hydrogen as a Carrier Gas
Introduction
Solvents are used across the whole chemical manufacturing industry, from pharmaceutical to pesticide production. The majority of solvents will be removed during the manufacturing process, however it is possible that residual levels of solvent will remain in the product.
Some solvents pose a risk to human health and therefore residual solvent analysis of technical grade ingredients is important before they are marketed to the public.
In this application note we will successfully validate a method for the analysis of residual solvents by GC-FID. A technical grade active ingredient (eugenol) will also be tested.
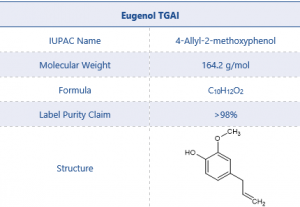
A commercially available sample of Eugenol was chosen as an example chemical product. Eugenol is a terpene which makes up around 80% of clove leaf oil and is attributed to the distinctive smell of cloves. Eugenol is also a powerful insecticide and when imported for this use falls under the requirements of the Plant Protection Products (PPP) or pesticide regulations.¹
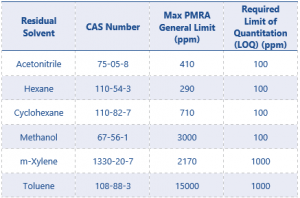
The government of Canada has published a list of generally acceptable limits of residual solvents in pesticides from the pest management regulatory agency (PMRA). Limits of solvents relevant to this application note can be found in Table 1.²
Table 1. List of generally acceptable limits of residual solvents in pesticides from the PMRA.²
Helium has long been the standard carrier gas for GC applications however, due to recent global shortages many companies are keen to find alternatives.³ The application described in this note has been performed using an alternative carrier gas, hydrogen. This can be produced with generators using renewable energy sources and so is a cleaner, greener option as well as avoiding the current issues over helium supply.³
Table 2. Test Item Details
This application can be performed on either the SCION Instruments 8300 GC or 8500 GC platforms equipped with an 8400 Pro Autosampler, S/SL injector, and FID detector.
Experimental
In lieu of testing guidelines SANCO guidance document SANCO/3030/99 rev5 was used due to the type of test item being used (eugenol).⁴
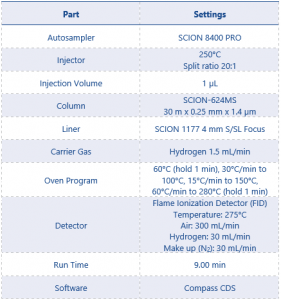
The instrument method parameters can be found in Table 3.
A suitable internal standard was chosen, limonene (purity 99.4%), which had a similar response factor to the target analytes and did not interfere with the active ingredient or any impurities present.
The internal standard solution was prepared by weighing 50mg limonene analytical standard into a 50mL volumetric flask, this was made to volume using DMF (HPLC grade, 99.9%) and mixed well.
Linearity samples were prepared as follows; a stock solution was prepared by weighing 25 mg of each solvent standard (Cyclohexane, Hexane, Acetonitrile and methanol) with a required limit of 100 ppm as per PMRA list into a 10 mL volumetric flask and making to volume with DMF. 125 mg of each solvent standard (toluene and m-Xylene) with a required ppm limit of 1000 ppm was weighed into a 5 mL volumetric flask and then made to volume with DMF. 0.5 mL of each standard solution was then pipetted into a 25 mL volumetric flask and made to volume with DMF to make the standard stock solution.
A number of dilutions were then performed by pipetting 0.25, 0.5, 1, 2, 3, and 4mL into separate 10mL volumetric flasks. 1 mL of the internal standard solution was added to each sample which were then made to volume with DMF.
Table 3. Instrument Parameters
A stock QC solution was prepared in the same manner as the linearity stock solution. From the QC stock solution 0.45 mL was pipetted into a 10 mL volumetric flask and made to volume with DMF.
SANCO defines precision as requiring a minimum of 5 determinations, we chose to prepare 6 samples. The low level recovery QC samples (×6) were prepared by weighing 200 mg of EGAI eugenol into a 10 mL volumetric flask and then spiking with 0.45 mL of the QC stock solution. 1 mL of internal standard was also added and the flask made to volume with DMF.
2 QC blank samples were also prepared to allow for standard addition method (subtraction of any solvent present in the technical material) by weighing 200 mg of eugenol into a 10 mL volumetric flask and making to volume with DMF and 1mL of internal standard solution. 2 technical samples were also prepared in the same fashion as the QC blank samples.
Results and Discussion

Figure 1. Example chromatogram for a QC sample
The instrument method stated in Table 3 gave excellent specificity. Figure 1 shows an example chromatogram of a QC sample.
This chromatogram clearly demonstrates the specificity of the method, showing excellent resolution between all the components as well as good peak shape of all residual solvents and the internal standard.
SANCO defines the criteria for acceptable linearity as a correlation coefficient (r²) value as >0.98. Table 4 shows the linearity results for all residual solvents.
Table 4. Summary correlation coefficient (r²) results for all residual solvents
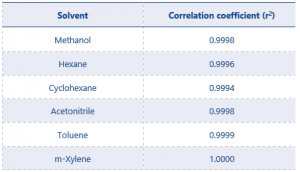
The r2 values of all solvents demonstrate excellent linearity, greatly exceeding the criteria defined by the SANCO document.
Precision results are determined to be acceptable using the Horwitz equation. The Horwitz equation corresponds to an exponential relationship between the estimated intra-laboratory repeatability (RSDr) and concentration (c).
![]()
For example the low QC for hexane was at a nominal concentration level of 0.0118% w/w which means the %RSDr or Horwitz value will be 5.23.
With this obtained value for the Horwitz value the Horwitz ratio (Hr) can be calculated, using the obtained repeatability (%RSD) of the target component.
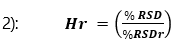
From this calculated value (Hr) the following criteria are applied:
Hr ≤ 1, acceptable.
1 < Hr ≤ 2, acceptable in case of a suggested explanation.
Hr > 2, not acceptable.
Assay accuracy and precision results are shown in Table 5 and 6.
Table 5. Assay Accuracy and Precision results for solvents at 0.01% w/w (LOQ determination)
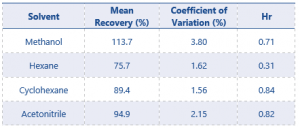
Table 6. Assay Accuracy and Precision results for solvents at 0.1% w/w
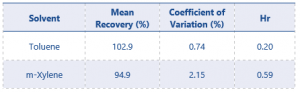
System precision results for all solvents can be found in table 7.
Table 7. System Precision results for all residual solvents
For results at the 0.01% w/w level recovery should be between 75 – 125%, and at the higher level recovery should be between 80 – 120% – these values have been greatly exceeded by the method.
The results for assay accuracy and precision easily pass the criteria set by SANCO. With these results the residual solvents method can now be considered fully validated and the content of residual solvents present in the product can be accurately quantified.
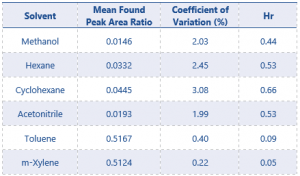
The only solvent found to be present in the test material was methanol. The results for residual solvent content in the test item can be found in Table 8.
Table 8. Results for residual solvent content in eugenol
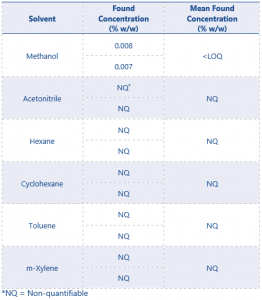
The 2 replicates of the blank QC samples that were fortified with eugenol product gave a mean found concentration of <LOQ for methanol. No other solvents were found to be quantifiable in the test material.
The residual solvents found in the test item were found to be well within the PMRA established limits discussed earlier.
Figure 1 shows the chromatogram of a QC sample.
Conclusion
Residual solvent analysis is important across a wide range of chemical industries, including the pesticide industry.
This application note has successfully validated a method for residual solvent analysis using a SCION GC-FID system, in compliance with the guidance document SANCO/3030/99 rev.5 for submission of a plant protection product or pesticide within the EU or UK.
The required limit of quantification set out by the PMRA was also met with the LOQ being determined at 100 ppm for acetonitrile, methanol, hexane and cyclohexane and the 1000 ppm limit was met for the m-xylene and toluene.
SCION instruments recommends checking with local regulatory authorities to ensure all testing and reporting requirements are met, or contact the SCION applications team for assistance.
References
1. Commission implementing regulation (EU) No 546/2013, Approval of Eugenol for use as a PPP, 14 June 2013, L 163/17
2. Government of Canada, https://www.canada.ca/en/health-canada/services/consumer-product-safety/pesticides-pest-management/registrants-applicants/established-limits-residual-solvents-technical-grade-active-ingredients.html, Accessed February 2024
3. Innovation News Network, Helium Shortage 4.0, Accessed January 2024, https://www.innovationnewsnetwork.com/helium-shortage-4-0-what-caused-it-and-when-will-it-end/29255/
4. European Commission, SANCO/3030/99 rev.5, 22 March 2019
Download Application Note
Download a PDF version of the application note which includes ordering information, using the following link: Residual Solvent Analysis of Chemical Products by GC-FID with Hydrogen as a Carrier Gas.
